Vitamin B12

by Jack Norris, RD
Contents
Essential Information
Vitamin B12 is arguably the most important nutrient in vegan nutrition. B12 is found in meat, dairy, and eggs. In contrast, there are no reliable plant sources of B12. Luckily, B12 doesn’t need to be obtained from animal foods because it’s made by bacteria. B12-fortified foods are widely available in many countries and B12 supplements tend to be common and inexpensive. The discovery of vitamin B12 in the 1940s allowed veganism to become a realistic lifestyle.
The overwhelming consensus in the mainstream nutrition community and among vegan health professionals is that B12 supplementation is necessary for the optimal health of vegans. The good news is that vegans who supplement with B12 often have a superior B12 status to meat-eaters.
But what if a vegan feels fine and so doesn’t bother supplementing with B12?
An overt B12 deficiency can develop, marked by fatigue or tingling in the hands or feet. In B12 deficiency, DNA synthesis in red blood cell precursors (erythroblasts) can be impaired, preventing the normal maturation and division of these cells, resulting in larger-than-normal red blood cells. This macrocytic or megaloblastic anemia can result in fatigue. B12 also protects the nervous system and without it someone can develop tingling in the hands or feet. If not treated, the deficiency can progress to more serious symptoms such as blindness, deafness, and dementia.
A less obvious B12 deficiency can also develop, marked by elevated homocysteine levels. Homocysteine is a potentially toxic byproduct of protein metabolism that the body clears with the help of B12. Elevated homocysteine levels have been consistently associated with an increased risk of cognitive decline, cardiovascular disease (especially stroke), and early mortality; there’s also a link with low bone mineral density. Many studies have found a high prevalence of elevated homocysteine among vegans who don’t supplement with B12. In contrast, vegans who supplement with B12 have healthy homocysteine levels. See Homocysteine and Mild B12 Deficiency in Vegans for more info.
A 1955 study from the U.K., one of the earliest studies of vegans, found a high prevalence of B12 deficiency with some vegans suffering from nerve damage and dementia. There have been many documented cases of individual vegans developing overt B12 deficiency symptoms after a period of not supplementing (see Individual Cases of B12 Deficiency in Vegans). And I’ve personally known numerous vegans who neglected B12 and subsequently experienced fatigue and temporary neuropathy that resolved upon supplementation.
If you look further into the details of vitamin B12, you’ll quickly find that they’re expansive and complicated. But one thing is simple: Vegans should ensure a reliable source of B12.
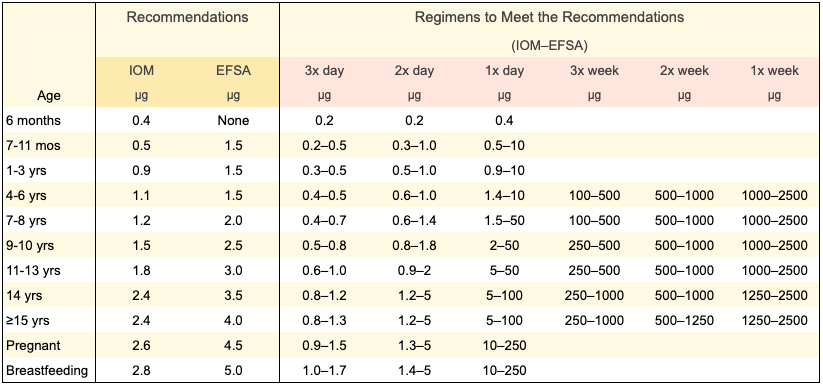
There are many options for meeting B12 recommendations, depending on how often you take B12 or eat fortified foods. The table below shows options for meeting the U.S. Institute of Medicine (IOM) recommended dietary intake or the European Food Safety Authority (EFSA) adequate intake. We explain how these amounts were calculated in our Rationale for Recommendations.
The recommendations above are for the cyanocobalamin form of B12 which is the form used for most fortified foods and many supplements. Cyanocobalamin is the form that we recommend because it’s more stable and has been researched more thoroughly than other forms. The hype around the other forms of B12 is mostly propaganda from the supplement industry. For more information on the forms, see Coenzyme Forms: Methylcobalamin and Adenosylcobalamin.
Additional Tips
- B12 is normally measured in micrograms, which can be abbreviated as either µg or mcg. 1,000 micrograms equals 1 milligram (mg).
- If you haven’t had a regular source of B12 for more than a few months, take 2,000 µg once a day for 2 weeks to replenish your stores.
- While there’s no definite cutoff for when larger amounts of B12 become unsafe, we somewhat arbitrarily recommend not taking more than 1,000 µg per day over the long-term without guidance from a physician.
- Large doses of B12 can trigger acne-like symptoms in a small number of people.
- To increase absorption, we recommend chewing B12-only supplements and swallowing multivitamins whole.
- People with kidney disease or a suspected B12 deficiency or malabsorption should talk to their doctors about an appropriate B12 regimen.
- In March of 2020, the Daily Value for B12 on food nutrition labels in the United States was lowered from the 1968 RDA of 6 µg to the current RDA of 2.4 µg.
- Store B12 supplements and fortified foods in a dark, cool place, preferably a refrigerator. Exposure to light can damage B12.
- B12-fortified toothpaste is available in some locations and has been shown to be a reliable source of B12 for vegans.
Further Information
- Can a Natural Diet Require Supplements?
- What Every Vegan Should Know about Vitamin B12: An Open Letter from Health Professionals and Vegan Organizations
Research on Vitamin B12
The links below provide a thorough review of the scientific literature on vitamin B12 and the vegan diet.
Rationale for VeganHealth’s B12 Recommendations
- Introduction
- Brief History of B12 Recommendations for Vegans
- Government Recommendations
- Cyanocobalamin Absorption
- Vegan Health B12 Recommendations
- Appendix A. Minimizing Methylmalonic Acid Levels
- Appendix B. Clinical Trials: Impact of B12 Supplementation on Metabolites
- Serum B12
- Holotranscobalamin
- Methylmalonic Acid
- Homocysteine
- Notes on Clinical Trials of B12 Supplementation
- Appendix C. Factorial Approach
- Appendix D. Pregnancy and Breastfeeding
- Appendix E. IOM Recommendations for People Over Age 50
- Appendix F. ASMBS B12 Recommendations for People after Bariatric Surgery
- Appendix G. Taking Vitamin B12 With or Without Food
- Bibliography
Are Multivitamins an Adequate Source of B12 for Vegans?
- Summary
- Introduction
- Clinical-Trials
- Observational Studies
- B12 Malabsorption
- Degradation of B12 under Experimental Conditions
- Yamada et al.’s 2008 Study of B12 in Fortified Beverages and Multivitamins
- Kondo et al. and Herbert et al.’s Research on Multivitamins circa 1980
- Potential Improvements in the Accuracy of Nutrition Labeling
- B12 Transport Proteins and Multivitamins
- Observational Study of B12 in Fortified Non-Dairy Milks
- Anecdotal Reports
- Dangers of Recommending “Take a Multivitamin”
- Conclusion
- Appendix A: Personal Experiment
- Bibliography
Elevated Vitamin B12 Levels and Mortality
Measuring B12: Why the Confusion?
- Summary
- B12 Amounts Versus B12 Activity
- Microbiological Assay
- R-protein Assay
- Intrinsic Factor Assay
- Intrinsic Factor Assay Shown to Be Unreliable in Humans
- Ochromonas Malhamensis Fares Better Than an Intrinsic Factor Assay
- Paper Chromatography
- Methods for Measuring B12 Activity of a Food
- Bacterial Contamination
- References
Vitamin B12 in Plant Foods
- Summary
- Introduction
- Plant Foods with Practically No Detectable B12 Analogue
- Fermented Foods
- Mankai (Duckweed)
- Algae
- Seaweeds (Macroalgae)
- Various Seaweeds: Dulse Warrants Further Study
- Nori
- Vitamin B12 Analogue Content of Nori Species
- Nori (Neopyropia sp.) Lowers Serum MMA Levels among Vegetarians (2024)
- Nori (P. tenera) Fails to Lower Urinary MMA Levels (1999)
- Coccolithophorid Algae
- Ulva Fenestrata
- A Case of False Reporting on the Benefit of Seaweed and Fermented Foods
- Genmai-Saishoku Paradox?
- German Whole Foods Vegans Consuming Nori and Mushrooms
- Soil and Organic Produce as a B12 Source for Vegans
- Bibliography
Subclinical B12 Deficiency and Homocysteine in Vegans
- Summary
- Background on Homocysteine
- Homocysteine in Vegetarians and Vegans
- Cognition
- B12 Status and Prospective Studies of Cognitive Decline
- B12 Metabolites and Brain Volume
- Randomized Controlled Trials of B-Vitamins, Cognitive Decline, and Brain Atrophy
- Research on Vegetarians and Vegans
- Summary of B12 and Cognition
- Mortality and Cardiovascular Disease
- Bone Mineral Density
- Bibliography

49 thoughts on “Vitamin B12”
Do you think a person who has some level of kidney disease without realising it might experience adverse effects from high dose cyanocobalamin?
How about someone who has previously had congenital kidney issues but has had that corrected, or else, someone with risk factors but no diagnosis? Would they need to be wary about possibly not clearing the cyanide?
With saying to replenish stores by taking 2,000 µg once a day for 2 weeks, does that also apply to children/teens who have been taking B12, but it has been found to be below the recommended dose?
And is there any risk of taking the high dose for a couple of weeks, without testing whether there is deficiency or not?
Ann,
I’m not aware of any evidence that 2,000 µg twice a day for 2 weeks is dangerous for children or teens. There are no upper limits set by the Institute of Medicine. However, the subject hasn’t been studied extensively. In adults, there are rare cases of cobalt sensitivity that appears as acne; these are thought to normally resolve upon ceasing the higher doses but it hasn’t been formally studied.
A prudent method would be to assume that the 2,000 mcg applies to someone weighing 150 lbs and to extrapolate to the weight of the child or teen for a lower dose (lower dose = 2,000 * child’s weight / 150) and then rounding to an amount that’s practical by either cutting the tablets in halves or quarters or buying smaller doses. And monitor for any facial changes. That probably sounds scary—it’s very rare, but still worth monitoring. Or, you can just opt for much lower, but not inconsequential, amounts like 100 µg/day. µg = mcg = microgram
Thanks, it’s difficult with teens also as they are expected to have acne.
How long do you think 100mcg per day would take to restore a deficiency for a child or teen?
The system for testing here seems to be that B12 and folate should be tested together, and if B12 levels are abnormal, then homocysteine is automatically checked. MMA testing may also be then funded. This seems not to take into account the way that if folate is adequate, it can mask low B12 symptoms and results, especially if MMA isn’t tested from the outset.
Do you think it’s possible to see from B12, folate and homocysteine results alone that B12 deficiency is being masked by folate?
Ann,
In my last response to you, I originally wrote “2,000 mg” but I meant “2,000 µg.”
> How long do you think 100mcg per day would take to restore a deficiency for a child or teen?
It should start to improve their B12 status within days. Are they having symptoms of B12 deficiency? How much do they weigh?
Thanks, not specifically B12 deficiency symptoms, but the multivitamin we were using turns out to have too low B12 (I originally checked it for a younger age bracket). Also it wasn’t being taken for a while as my child stopped liking it and we tried a few brands before finding another one, which is now 3mcg per tablet, 3 times a day (as the packet says 2-3 per day, but I’m separating the times).
Even the nutritional yeast I was buying, which says ‘source of B12’ on the packet, turns out to have just 3.3ug per 100g.
So there has been undersupplementation for a while; no tingling or balance issues, but I also read that folate can mask some symptoms of B12 deficiency till it’s too late to reverse damage. Giving a deficiency dose for a while seems possibly a good idea. Based on the equation in your comment, 500mcg could be ok.
I just spoke with the doctor and they recommended 100-200mcg per day for 3-6 months. I guess 100mcg twice a day isn’t the intention there; it may be ok to stay with 200mcg once per day? (I could talk with the doctor again if needed.)
Thanks.
Ann,
> Also it wasn’t being taken for a while as my child stopped liking it and we tried a few brands before finding another one, which is now 3mcg per tablet, 3 times a day (as the packet says 2-3 per day, but I’m separating the times).
That’s plenty of B12 for a child or an adult.
Folate can’t mask the neurological problems caused by B12 deficiency; it can only mask the red blood cell symptoms.
Your doctor’s advice sounds reasonable.
> Also, I read that sublingual B12 isn’t necessarily better than normal tablets, and intrinsic factor does most of the absorption. Do you know if that recommendation has changed?
No, you don’t need sublingual.
The amount of cyanide is physiologically insignificant and the precautions for people with kidney disease are just to be extra careful, not because of any cases of definite kidney damage. But there’s methylcobalamin instead of cyanocobalamin; I’d recommend talking to your doctor about it.
Also, I read that sublingual B12 isn’t necessarily better than normal tablets, and intrinsic factor does most of the absorption. Do you know if that recommendation has changed?
Thanks. The kidney question is regarding someone else, good to know that the risk doesn’t seem so much.
I wonder if 3x 3mcg per day is similar to 1x 100mcg, in terms of what is absorbed. If a lower supplement amount can have the same impact and possibly less chance of skin imbalances, maybe that’s preferable.
I also read that high B12 supplementation can cause acne because skin bacteria stop producing B12 and start producing more porphyrins. If that’s true, I wonder how it can be distinguished from cobalt sensitivity.
Ann,
The supplemental amounts in the rows of this chart were devised to provide approximately the same amounts of absorbed B12.
> I also read that high B12 supplementation can cause acne because skin bacteria stop producing B12 and start producing more porphyrins. If that’s true, I wonder how it can be distinguished from cobalt sensitivity.
That appears to be true (according to this study) but it probably applies mostly to people receiving injections. Someone who receives a vitamin B12 injection of 1,000 µg will effectively “absorb” all 1,000 µg while someone taking a 1,000 µg oral supplement will only absorb about 1%. Only 1 out of 10 healthy subjects receiving an injection developed signs of acne.
I’ve been reading a lot about folate and folic acid the past few days and found out something that had slipped past my attention previously—that food folate can’t mask a B12 deficiency, only supplemental folic acid, 5-MTHF-glucosamine, and L-5-MTHF-Ca can mask B12 deficiency. It’s kind of the key point of the methylfolate trap, and I’m a little embarrassed to say that I never realized it. I’ve updated the section of the site about folic acid masking B12 deficiency.
The wording is off when you say, “B12 is normally needed for red blood cells to divide and become active-“. Red blood cells don’t have a nucleus or DNA and therefore cannot divide. Please update the wording to read, B12 is needed for red blood cell precursors or something along those lines.
Dustin,
Thank you. I tried to clean that section up.
Thank you for this information. It was a tad too scientific for my little brain so I did skim a lot. I will not ask the questions you say that you will not answer but I hope you answer the question I do ask. It regards the dosage. The bottles I see for sale offer B12 in crazy excesses. Why is that? I have a bottle of 500 mcg which is 20833 % of the RDA. I have another bottle of 25 mcg which is 1042% of the RDA. If they have established an RDA, why not 100% of it instead of these huge amounts? Your article said that the body could store this for up to 2 yrs–somebody else told me that the body excretes the excess. Is it stored or is it excreted? Why do they sell such huge doses? Thanks a lot.
Martha,
When you ingest B12, some of it is absorbed and some of it is excreted (through your feces). Most of the B12 that is absorbed will be stored until it’s used. Did my article say it could be stored for 2 years? I don’t remember saying that. But there’s no set amount of time for which any given molecule of B12 is stored.
Here’s why the doses are so high:
I’m excerpting from the article, Rationale for VeganHealth’s B12 Recommendations:
> Government recommendations for B12 are created with the assumption that people receive multiple doses of B12 from foods throughout the day. In typical dietary doses, B12 is absorbed at a rate of about 40-50%. Intrinsic factor, a protein that shuttles B12 through the absorption process, becomes saturated after a meal containing an above-average amount of B12 and about 4 to 6 hours are required before it can efficiently absorb another dose (IOM, 1998). However, a very small percentage of B12 from a supplemental dose is absorbed by passive diffusion, rather than by intrinsic factor, such that vegans can make up for taking B12 only once per day, or even only once per week, by taking larger doses than normally provided by food.
B12 recommendations, such as the RDA, should be thought of as based on how much B12 is absorbed rather than how much is ingested. The less frequently someone takes B12, the more they need in order to absorb enough to meet the recommendations. If you take B12 3 times a day, your total amount needs only to meet the RDA. But, if you take it, for example, only once a week, you need much higher doses.
I’ve been taking B12 for years as soon as I started a vegan diet. I took test two times and the results were good. At that time I was not consuming any seaweed which could interfere with the results. Now I started experimenting with low daily doses of B12, however with methylcobalamin, since cyano form does not dissolve in water at all. I take 25 mcg of MeCbl solution two times a day with food, with hours of interval. I believe that should be enough to saturate intrinsic factor fully and reach the DR of B12. Should I get checked on B12 levels after some time to see if this method works good enough, or there should be no problem?
Hi Sandro – We can’t give advice on each person’s personal B12 regimen, but check out this article: https://veganhealth.org/should-i-get-my-b12-status-tested. And here are our recommendations: https://veganhealth.org/daily-needs/#Vitamin-B12.
Dear Jack and colleagues! Could you please comment on this study: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6642017/
Seems you didnt review it in Elevated B12 section. It supports the idea that high B12 levels _cause_ elevated lung cancer risk.
Hi Nikita,
Here are my comments: Elevated B12 and Lung Cancer
The information in here is not true, and not well researched. There are plenty of vitamin b12 sources that are vegan. Sure, some seaweed has little to no b12, but not all. 2.5 grams of dried Pyropia yezoensis has over 35% of the RDI of b12. Fermented foods, however are by far the best source of vitamin b12 for vegans. One 10oz glass of kombucha contains over 340% of the RDI for b12. There’s absolutely no need for b12 supplements, and I’m inclined to believe the only reason you are saying this is because you have some sort of monetary incentive to.
Please see this article: https://veganhealth.org/vitamin-b12-plant-foods/. We’re unaware of any published research showing that kombucha contains B12.
Hi there,
very interesting post, thanks for collecting all this info! I recently began researching about B12 sources. I found two studies which show that Chlorella vulgaris is a source of active cobalamin. Specifically, supplements of C. vulgaris apparently contains both cyanocobalamin and metylcobalamin, as determined per HPLC analyses [1]. Also, C. vulgaris supplements had activity reducing the risk of anemia, proteinuria and edema in pregnant women [2]. Since B12 can only be produced by prokaryotes, C. vulgaris must obtain B12 from the media which is produced by bacteria or archaea. Thus, I think that it is worth considering C. vulgaris supplements as sources of active cobalamin. What do you think?
Best,
Semidan
[1] N. Jaliliana, G. D. Najafpour and M. Khajoueib, 2019. Enhanced Vitamin B12 Production using Chlorella vulgaris
[2] Shiro Nakano, Hideo Takekoshi and Masuo Nakano, 2010. Chlorella pyrenoidosa supplementation reduces the risk of anemia, proteinuria and edema in pregnant women
We have written about chlorella and B12 here: https://veganhealth.org/vitamin-b12-plant-foods/#chlorella. Until chlorella is shown to consistently reduce MMA levels in humans (we are unaware of studies showing this), we cannot recommend it as a source of vitamin B12.
I recently found a B-Complex product that appears to be sourced from quinoa sprouts. The product includes B-12, but I’m concerned that this may be an inactive analogue unless they are just using the quinoa as a medium for bacteria-derived B-12. The product is produced by Swanson, and when I look at the Supplement Facts on the back, each vitamin lists next to it in parentheses “PANMOL(r) B-COMPLEX from quinoa sprouts.”
There’s very little information on the PANMOL site, and the research section does not list any actual research. I’m guessing they sell directly to supplement manufacturers, but this is unclear as well. They’re an Austrian company, but it looks like they partner with a company in the USA called Stauber. The Swanson product I bought is vegan, but it looks like the Stauber product may not be, but that’s neither here nor there since I’m just looking for info about the PANMOL itself. The Stauber site claims that all of the B vitamins are in their active forms.
I’m having a hell of a time trying to find actual research on this. Do you think this is a B-12 analogue or actual biologically-active B-12. I’m concerned that I may have purchased an ineffective product. My Google searches have found nothing on B-12 from quinoa sprouts either. Thanks for your help!
Meredith,
If “cyanocobalamin” isn’t listed in the ingredients, then I wouldn’t rely on it as a sole source of vitamin B12.
Thanks! I managed to track down the FDA GRAS Notification for PANMOL. It just says “cobalamine” in the document but not which kind, so I will be looking for a different supplement.
Hi Meredith- were you able to find out if PANMOL contains cyanocobalamin OR methylcobalamin? Are you still using the product you mentioned?
So now we have a VIABLE Plant Based Source of B-12 that is literally part of the plant’s biomass. Water Lentils AKA Duckweed.
We looked into duckweed and determined it is much too soon to conclude that it is a reliable source of vitamin B12 for humans.
What’s with the new study about high B12 intake and hip fractures (in older women)? I am in particular taken aback by the extreme discrepancy between what the study labels high intake (20 mcg of B12) and the daily dosage of supplement I take (500 mcg).– an older man
Given that the findings from this study on B12 and hip fractures are statistically weak, that they’re based on an observational study (which only shows correlations), and go against research showing a beneficial effect of higher B12 levels on bones, we don’t think this evidence is enough to justify changing our recommendations. While we generally recommend 500 µg/day for people over 65 (because of the research explained here), if you get your B12 levels tested regularly and they’re within the normal range, it would appear safe to follow the recommendations for people under 65.
Duckweed – Have you heard that there is evidence that it contains active B12?
Research sounds promising
We looked into duckweed and determined it is much too soon to conclude that it is a reliable source of vitamin B12 for humans.
We added information about duckweed here: https://veganhealth.org/vitamin-b12-plant-foods/#duckweed
As well as a blog post: https://veganhealth.org/is-duckweed-a-source-of-vitamin-b12/
I am vegan because I care for nonhuman animals who are mistreated by humans. Their lives are worth the same, if not more that humans, therefore I won’t take anything that comes from them. It is just not ethical. I already have lots of benefits from being vegan: lots of energy; strong immune system, I do not get cold or flu even if I am exposed to it or I’ll get immunity by having a sub clinical illness which gives me immunity against that specific strain; if I get a cut or another injury, I heal fast. Another benefit I get is my risks for cancer and cardiovascular disease are reduced.
Since animals make vitamin B12 in their system, is there an specific microbial source that can strengthen my own bacteria producing B12?
Hi Norma, the place in our intestines where bacteria make vitamin B12 is lower down than where we absorb it. Which means that we do not absorb vitamin B12 made by the bacteria in our intestines. The only reliable vegan sources of vitamin B12 are fortified foods and supplements.
A vegan mom since pregnancy is low on breastmilk and feeding her 5 mos old a vegan formula . This homemade formula is Almond milk , spirulina, nutritional brewers yeast, sacha,powders, coconut oil are main ingredients . Can an baby aborsorb vita b 12 from such a mix?
Please see the Pregnancy, Infants, and Children section of our website for more information and resources for vegan infants. Specifically, this section says:
“Vegan parents should not try to make their own infant formulas as this often leads to poor child development. Although more research is desirable, it appears that soy infant formulas are safe. See below for information regarding soy formulas.”
In addition to real concerns about vitamin B12 from the formula you describe, calories, protein, calcium, iron, zinc and many other nutrients could be too low or too high. Please, for the sake of this infant’s health and well-being, strongly encourage this mom to change her plans. Some suggestions:
• consult a certified lactation specialist to address breastfeeding issues
• use a commercial soy formula in place of the homemade formula
• consult with a registered dietitian to discuss feeding vegan infants and children.
Is there any reason to avoid the cyano form when breastfeeding infants, in case maybe it could burden their young liver, or any other reason?
Cyanocobalamin is used in infant formulas so it is likely that this form can be used by infants.
Hi. I am obese and trying to eat healthier. Went on a 10 day whole plant based diet through a work-wide health challenge and while I don’t envision adopting this diet for life, I would like to try and use the recipes I like and try to avoid dairy and oil while limiting meat consumption to poultry now and then. I started taking B12 with the challenge (I’m on day 15). I had no idea what to buy. My bottle says 3000 mcg and directions say to take one daily. Please let me know if this is too much. Also, I am diabetic, on medications, and have blood work taken evey 6-8 months. My doctor has never said anything about my vitamin B12. He did put me on vtiamin D3, 2000 units, daily. I would appreciate your advice. Would you please email your response as I found this website while scrolling and not sure I’ll get back! Thank you very much.
According to the National Institutes of Health, (https://ods.od.nih.gov/factsheets/VitaminB12-Consumer/#h8) vitamin B12 can interfere with some medications including a drug used to treat diabetes. It’s important to tell your health care provider about any supplements that you take.
Hi I’m having much acne since I’m taking cyanocobalamin pills. I have stopped taking them as a temporary measure. I am vegan and I don’t know what I should do in the long term. Any advice? I’m quite worried. Thank you in advance!
Belen,
B12 fortified foods or a multivitamin or supplement with much lower amounts of B12. See the “Option 1” column in the B12 table here: https://veganhealth.org/daily-needs/
What’s your opinion regarding taking B12 individually or as part of a “B complex” ie. with several B-vitamins? Does the latter increase absorption?
Marcus,
I’m not aware of any evidence that taking B12 as part of a vitamin B complex increases its absorption.
I mean this article is missing (got from web arhive) https://web.archive.org/web/20170505225006/http://veganhealth.org/b12/inc
Nikita,
I took that page down when I migrated everything to the new site because it was so outdated and due to the fact that spurious correlations between disease and high B12 levels are too burdensome to track just to say we did it. If any research comes out that seriously implicates high B12 intakes as a cause of disease, we’ll cover it on our updates blog which you can sign up for at the bottom of each page on the website.
Seems that the old site article “Elevated Serum B12 and Increased Risk of Disease” dissapears. Can’t find it… Did you remove it? Why?
Lots of information and still looking for a clear answer.. Do we need! B12 supplements as vegan, vegetarian or meat eater?
Bernadette,
Vegan or near-vegan: yes, see Daily Needs
Someone who eats animal products multiple times a day (in other words NOT a vegan or near vegan): no